Volvo goes green(er) with its big vehicle approach to environmentalism. When I say big, think eighteen-wheeler-big. The Swedish company's truck division is testing hydrogen-powered heavy haulers on public roads.
The fuel-cell-electric machines have undergone rigorous trials on closed roads. The manufacturer now kicks it up a notch. The massive vehicles must prove their worth in real-life conditions to pull ahead (pun intended) toward a green future.
Swedes are known to have an original approach to motoring – and Volvo is a name that needs no introduction to any gearheading congregation. For comparison, another company makes cars and bears the same nationality (Koenigsegg). Vastly different in philosophy, the two share a no-corners-cut point of view regarding the transfer of technology to their road-going creations.
The famous brand that put the three-point seatbelt on the roadmap to safety is now foreseeing a water-byproduct future of automotive. The fuel cells use fundamental chemistry to make electricity: combine hydrogen and oxygen, extract energy from the reaction, and eliminate the residues.
Elementary school science class taught us all that hydrogen + oxygen = water + heat. Unless Volvo has a go at it and utilizes a fuel cell to optimize the basic formula and extract electricity from the mixture. From a strict engineering perspective, this cycle is virtually endless – the resulting water is dispersed into the atmosphere.
At one point or another, water is subjected to electrolysis and deconstructed into its primary constituents to obtain hydrogen and oxygen to power the fuel cells. Those emit water, and that's then… you get the point. Volvo Trucks put their most energy-intensive vehicles under test – trucks that rely on electricity produced by hydrogen fuel cells.
Because they are Swedes, they didn't just send the vehicles on a B road somewhere in the country's remote north. They specifically chose the worst possible environment – the arctic winter – and went full ballistic (watch the first video for details).
Liquid hydrogen is a very volatile and aggressive fuel. It requires very strict storage conditions and materials to deliver a high-efficiency reaction. This, in turn, is needed to create enough electric power to move a truck through snow, cold, and blizzard-level winds.
The Swedish logic is bullet-proof: if the fuel cell technology is feasible close to the North Pole, it can be used anywhere (or beyond this cosmic boundary). Following the success of this second testing phase, Volvo intends to make the technology commercially available in the second half of this decade.
Whether that means 2025 or 2029 is a matter of fine-tuning engineering (pun intended) to improve this alternative to ICE and battery-electric powertrains. But before the mass-scale launch, hydrogen-electric trucks will undergo a third testing stage – with various haulers around the globe.
To make development as fast as possible, Volvo turned to Daimler to develop and produce fuel cell systems specifically for long-range heavy-duty vehicles. Volvo's hydrogen-powered trucks will use two fuel cells with a combined generating capacity of 300 kW of electric power.
A fuel cell is – in a way - a middle-of-the-road technology between classic internal combustion and full-electric power sources. It resembles the hydro-carbon-burning engine in the sense that the fuel cell also makes use of a chemical reaction to generate energy. But it differs because it doesn't burn a fluid (either a liquid or a gas) to turn it into heat and residues.
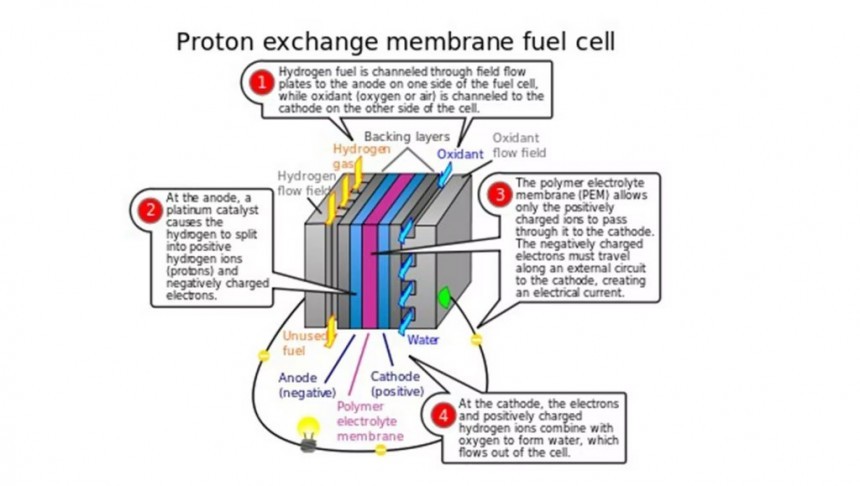
Instead, liquid hydrogen stored in a pressurized tank is fused with oxygen to produce electricity. The process occurs in a PEM (Polymer Electrolyte Membrane) stack composed of thin, thin plates separated by membranes.
The diagram above this paragraph is a far more accurate - albeit basic - description of how the fuel cell functions. Also, play the second video attached to this story for a brief introduction to hydrogen electricity. Although it is relatively less economically efficient than the classic diesel alternative, hydrogen-electric technology is still in its infancy.
And let's remember how long a way BEVs have come in the last decade alone. Hydrogen fuel cell technology has all the correct arguments to match, if not surpass, today's sweetheart solution for clean motoring. After all, hydrogen is only our planet's most abundant chemical element. And in our Universe, too.
Apart from fuel cell technology, hydrogen can also be used identically to diesel or gasoline in an internal combustion engine. Instead of injecting one of the two flammable liquids into the cylinder heads, hydrogen is forced to react with oxygen, creating the famous controlled explosion that pushes the piston down.
This solution is less efficient than fuel cells because internal combustion hydrogen needs to stay liquid. It is currently very challenging to keep it in that state for more than a few hours. On a positive note, liquified hydrogen doesn't require ultra-strong high-pressure tanks for storage. It is also more energy-dense than the gaseous alternative.
However, apart from being very flammable – and extremely prone to catastrophic explosions – hydrogen is also highly corrosive (again, remember that the most aggressive acids known to chemistry are hydrogen-and-something-else compounds). While this feature can and is kept under control, it is pretty costly. The installations used in producing, storing, and transporting hydrogen are either made of special materials – meaning high costs – or need to be replaced often (and that's another pocket-hole drill).
Swedes are known to have an original approach to motoring – and Volvo is a name that needs no introduction to any gearheading congregation. For comparison, another company makes cars and bears the same nationality (Koenigsegg). Vastly different in philosophy, the two share a no-corners-cut point of view regarding the transfer of technology to their road-going creations.
The famous brand that put the three-point seatbelt on the roadmap to safety is now foreseeing a water-byproduct future of automotive. The fuel cells use fundamental chemistry to make electricity: combine hydrogen and oxygen, extract energy from the reaction, and eliminate the residues.
At one point or another, water is subjected to electrolysis and deconstructed into its primary constituents to obtain hydrogen and oxygen to power the fuel cells. Those emit water, and that's then… you get the point. Volvo Trucks put their most energy-intensive vehicles under test – trucks that rely on electricity produced by hydrogen fuel cells.
Because they are Swedes, they didn't just send the vehicles on a B road somewhere in the country's remote north. They specifically chose the worst possible environment – the arctic winter – and went full ballistic (watch the first video for details).
The Swedish logic is bullet-proof: if the fuel cell technology is feasible close to the North Pole, it can be used anywhere (or beyond this cosmic boundary). Following the success of this second testing phase, Volvo intends to make the technology commercially available in the second half of this decade.
Whether that means 2025 or 2029 is a matter of fine-tuning engineering (pun intended) to improve this alternative to ICE and battery-electric powertrains. But before the mass-scale launch, hydrogen-electric trucks will undergo a third testing stage – with various haulers around the globe.
A fuel cell is – in a way - a middle-of-the-road technology between classic internal combustion and full-electric power sources. It resembles the hydro-carbon-burning engine in the sense that the fuel cell also makes use of a chemical reaction to generate energy. But it differs because it doesn't burn a fluid (either a liquid or a gas) to turn it into heat and residues.
Instead, liquid hydrogen stored in a pressurized tank is fused with oxygen to produce electricity. The process occurs in a PEM (Polymer Electrolyte Membrane) stack composed of thin, thin plates separated by membranes.
And let's remember how long a way BEVs have come in the last decade alone. Hydrogen fuel cell technology has all the correct arguments to match, if not surpass, today's sweetheart solution for clean motoring. After all, hydrogen is only our planet's most abundant chemical element. And in our Universe, too.
Apart from fuel cell technology, hydrogen can also be used identically to diesel or gasoline in an internal combustion engine. Instead of injecting one of the two flammable liquids into the cylinder heads, hydrogen is forced to react with oxygen, creating the famous controlled explosion that pushes the piston down.
However, apart from being very flammable – and extremely prone to catastrophic explosions – hydrogen is also highly corrosive (again, remember that the most aggressive acids known to chemistry are hydrogen-and-something-else compounds). While this feature can and is kept under control, it is pretty costly. The installations used in producing, storing, and transporting hydrogen are either made of special materials – meaning high costs – or need to be replaced often (and that's another pocket-hole drill).