Researchers at Tohoku University of Sendai (Japan) have found a way to stabilize lithium depositions in Li-Ion batteries, which helps prevent dendrite formation and capacity degradation. Their research also works for sodium batteries, paving the way for higher energy density batteries with extra-long lifecycles.
Li-Ion battery research is far from over because the technology is far from perfect. Even the most advanced batteries have shortcomings, depending on their chemistry and packaging. The goal of having safer, higher-capacity, faster-charging, and longer-lifecycle batteries remains a dream, at least for now. Scientists are working hard to improve existing battery designs while finding new chemistries and processes to push the envelope for the above qualities.
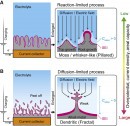
Currently, the most promising battery designs use alkali metal anodes, lithium and sodium being among the preferred choices. In Li-ion batteries, ions pass from the cathode to the anode when charging and in the opposite direction when the battery generates power. While doing so, metal is repeatedly deposited and dissolved, leading to an uneven structure of the lithium anode. This degrades the battery capacity, which would become unusable after a finite number of charge/discharge cycles.
The same process also leads to the formation of needle-like structures on the electrodes called dendrites. This can puncture through the isolator layers, causing short-circuiting. In the worst cases, the battery overheats and catches fire. In milder cases, it will further reduce battery capacity.
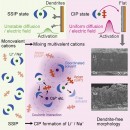
Fortunately, researchers at the Tohoku University’s Institute for Materials Research have found a way to eliminate this problem. They discovered that adding multivalent cations, such as calcium ions, to the electrolyte can reduce and even eliminate the dendrite formation.
“Our modified structure moderates the reduction of lithium or sodium ions on the electrode surface and enables a stable diffusion and electric field,” said Dr. Hongyi Li, the team leader of this research. In turn, the stabilized ions preserve the electrodeposited metals’ structure.
The process is, of course, more complicated than that. Those who want more in-depth information can go to the ScienceDirect website and read the scientific research published in the Cell Reports Physical Science journal. For others, it’s good to know that the researchers are working now on improving the metal anodes’ interfacial design. This should further enhance the cycle life and power density of the batteries.
Currently, the most promising battery designs use alkali metal anodes, lithium and sodium being among the preferred choices. In Li-ion batteries, ions pass from the cathode to the anode when charging and in the opposite direction when the battery generates power. While doing so, metal is repeatedly deposited and dissolved, leading to an uneven structure of the lithium anode. This degrades the battery capacity, which would become unusable after a finite number of charge/discharge cycles.
The same process also leads to the formation of needle-like structures on the electrodes called dendrites. This can puncture through the isolator layers, causing short-circuiting. In the worst cases, the battery overheats and catches fire. In milder cases, it will further reduce battery capacity.
Fortunately, researchers at the Tohoku University’s Institute for Materials Research have found a way to eliminate this problem. They discovered that adding multivalent cations, such as calcium ions, to the electrolyte can reduce and even eliminate the dendrite formation.
“Our modified structure moderates the reduction of lithium or sodium ions on the electrode surface and enables a stable diffusion and electric field,” said Dr. Hongyi Li, the team leader of this research. In turn, the stabilized ions preserve the electrodeposited metals’ structure.
The process is, of course, more complicated than that. Those who want more in-depth information can go to the ScienceDirect website and read the scientific research published in the Cell Reports Physical Science journal. For others, it’s good to know that the researchers are working now on improving the metal anodes’ interfacial design. This should further enhance the cycle life and power density of the batteries.