When people ask who came first, the chicken or the egg, the answer is so evident that the question got outdated. Eggs existed way before birds emerged: first with fish, then with reptiles. Apply that to FCEVs (fuel cell electric vehicles), and the question gets more interesting: what will come first, FCEVs or a hydrogen network? A cheaper fuel cell can help solve that question, and Cornell University researchers may have found a way to deliver them.
Fuel cells are great because they make hydrogen react with oxygen and generate electricity in the process while delivering only water as a byproduct. One of the key elements in a fuel cell is platinum. This precious metal is used to catalyze ORR (oxygen reduction reaction), the most crucial reaction not only in fuel cells but also in breathing. As it is normally very slow, platinum helps it occur faster. The problem is that platinum is costly. To make matters worse, Russia is one of the leading producers of this metal.
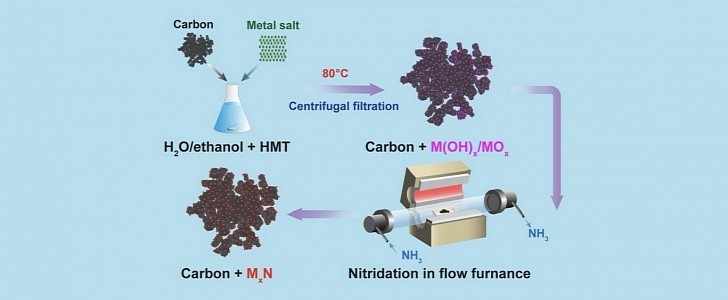
What these Cornell University researchers discovered was a catalyst for ORR that was 475 times cheaper than platinum when the study was published on February 2, 2022. With Putin and his supporters attacking Ukraine, the price differences have certainly changed, and we’d bet they are even more significant.
These nonprecious metal derivatives are called TMNs (transition metal nitrides), and the researchers made them from manganese, iron, and cobalt. According to the lead author Héctor D. Abruña, the cobalt nitride catalyst was “the clear winner." Sadly, cobalt is not a metal readily available or free of controversies, as battery manufacturing demonstrates.
The catalyst material the researchers developed works just fine with alkaline fuel cells. They are one of the most promising among the five main types – the others are PEM (proton exchange membrane), PAFC (phosphoric acid fuel cell), MCFC (molten carbonate fuel cell), and SOFC (solid oxide fuel cell). Alkaline fuel cells work at ambient temperature and have an efficiency of up to 70%.
If fuel cell manufacturers manage to adopt cobalt nitride catalysts – and produce them in enough numbers to replace the platinum – they will get a lot less expensive. That would favor the production of these fuel cells at competitive prices to adopt them in mass-production vehicles. With demand, we would probably see more hydrogen stations around the world.
We’d love to see them used in plug-in hybrids fed by fuel cells instead of combustion engines. They could have battery packs big enough for an intermediary range (more than 100 kilometers, or 62 miles) that allowed people to drive daily with no need for hydrogen. Hydrogen stations placed on highways (mainly for trucks) could provide the gas for passenger cars in long-distance travels.
The same war that is pushing gas and diesel prices to new highs might end up creating the necessary financial stimulus for a hydrogen infrastructure to be established. That would make the chicken emerge before the eggs, pushing them to multiply (no pun intended). We’d skip war to get there if that was possible. If there is any bright side to all this, it probably is that we should get rid of the fossil fuels dictators and autocrats love so much to sell as soon as possible. The Cornell University research may help us get there.
What these Cornell University researchers discovered was a catalyst for ORR that was 475 times cheaper than platinum when the study was published on February 2, 2022. With Putin and his supporters attacking Ukraine, the price differences have certainly changed, and we’d bet they are even more significant.
These nonprecious metal derivatives are called TMNs (transition metal nitrides), and the researchers made them from manganese, iron, and cobalt. According to the lead author Héctor D. Abruña, the cobalt nitride catalyst was “the clear winner." Sadly, cobalt is not a metal readily available or free of controversies, as battery manufacturing demonstrates.
The catalyst material the researchers developed works just fine with alkaline fuel cells. They are one of the most promising among the five main types – the others are PEM (proton exchange membrane), PAFC (phosphoric acid fuel cell), MCFC (molten carbonate fuel cell), and SOFC (solid oxide fuel cell). Alkaline fuel cells work at ambient temperature and have an efficiency of up to 70%.
If fuel cell manufacturers manage to adopt cobalt nitride catalysts – and produce them in enough numbers to replace the platinum – they will get a lot less expensive. That would favor the production of these fuel cells at competitive prices to adopt them in mass-production vehicles. With demand, we would probably see more hydrogen stations around the world.
We’d love to see them used in plug-in hybrids fed by fuel cells instead of combustion engines. They could have battery packs big enough for an intermediary range (more than 100 kilometers, or 62 miles) that allowed people to drive daily with no need for hydrogen. Hydrogen stations placed on highways (mainly for trucks) could provide the gas for passenger cars in long-distance travels.
The same war that is pushing gas and diesel prices to new highs might end up creating the necessary financial stimulus for a hydrogen infrastructure to be established. That would make the chicken emerge before the eggs, pushing them to multiply (no pun intended). We’d skip war to get there if that was possible. If there is any bright side to all this, it probably is that we should get rid of the fossil fuels dictators and autocrats love so much to sell as soon as possible. The Cornell University research may help us get there.