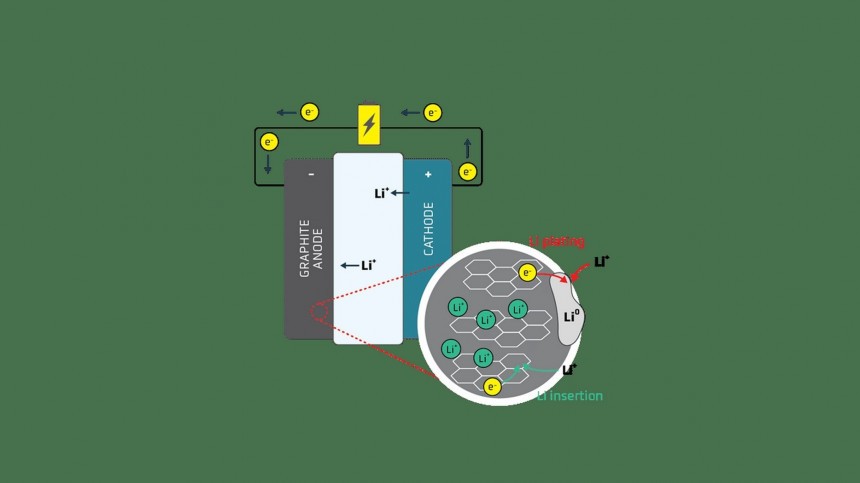
Solid-state Li-ion batteries are considered the next big thing for electric vehicles, but they form mysterious, branch-like metallic filaments, called dendrites, that can quickly compromise them. MIT researchers have found what causes them and how they can be prevented.
Replacing the liquid electrolytes in Li-ion batteries with solid matter can elevate them to a new level of safety and performance. Solid-state batteries, as they are called, are more lightweight, compact, and safe. Still, they also suffer from a mysterious problem that prevents them from being viable solutions for powering an EV.
This problem is caused by dendrites, branch-like filaments that can build up on the lithium surface and penetrate the solid electrolyte. Eventually, the dendrites (a name that comes from the Latin for branches) can cross from one electrode to the other, shorting out the battery cell. This essentially means its demise, but since Li-ion batteries pack a lot of energy, it can be an explosive event, negating all the safety promised by using a solid electrolyte.
You can bet that researchers around the world have been busy trying to figure out what causes dendrite formation and how it can be prevented, to no avail. New research published by MIT professor Yet-Ming Chiang and his team in the journal Joule seems to solve the old problem of dendrite formation. It not only explains how they form, but also how dendrites can be prevented from crossing through the electrolyte.
Like many discoveries in human history, this was a “surprising and unexpected” finding. Many people wondered how it was possible for the hard, solid electrolyte material used for a solid-state battery to be penetrated by lithium, which is a very soft metal. Apparently, this is made possible by the cracks that appear in its structure during charging and discharging. These cracks are not caused by a purely electrochemical process, as was previously thought. Instead, it’s a mechanical process that causes the problem.
Since the process of dendrite formation takes place deep within the opaque materials of the battery cells, it cannot be directly observed. The team developed a transparent electrolyte, allowing the whole process to be directly seen and recorded. Using the knowledge they gathered, the team members demonstrated that they could directly manipulate the growth of dendrites simply by applying and releasing pressure, causing the dendrites to zig and zag in perfect alignment with the direction of the force.
Applying mechanical stress to the solid electrolyte doesn’t eliminate the dendrite formation, but can control the direction of its growth. Instead of allowing them to cross from one electrode to the other, the MIT researchers proved they could be directed to remain parallel to the two electrodes. Thus, they remain harmless, allowing the solid-state cells to endure many charge-discharge cycles without shorting out.
The team found that bending the electrolyte can exert pressure and prevent dendrites from crossing. In practice, there could be better solutions, such as using two layers of material with different amounts of thermal expansion. This causes an inherent bending of the material, as is done in some thermostats. Another approach is to dope the electrolyte with atoms that would become embedded in it, distorting it and leaving it in a permanently stressed state. This method is used to produce super-hard glass for smartphones.
Having demonstrated the theoretical principles of this process, the team will try to apply them to a functional battery prototype. Although a patent has been filed, the MIT researchers don’t intend to commercialize the system themselves. Instead, they think the companies working on the development of solid-state batteries should benefit from their findings. Hopefully, this will lead to putting solid-state batteries on the market as soon as possible.
This problem is caused by dendrites, branch-like filaments that can build up on the lithium surface and penetrate the solid electrolyte. Eventually, the dendrites (a name that comes from the Latin for branches) can cross from one electrode to the other, shorting out the battery cell. This essentially means its demise, but since Li-ion batteries pack a lot of energy, it can be an explosive event, negating all the safety promised by using a solid electrolyte.
You can bet that researchers around the world have been busy trying to figure out what causes dendrite formation and how it can be prevented, to no avail. New research published by MIT professor Yet-Ming Chiang and his team in the journal Joule seems to solve the old problem of dendrite formation. It not only explains how they form, but also how dendrites can be prevented from crossing through the electrolyte.
Since the process of dendrite formation takes place deep within the opaque materials of the battery cells, it cannot be directly observed. The team developed a transparent electrolyte, allowing the whole process to be directly seen and recorded. Using the knowledge they gathered, the team members demonstrated that they could directly manipulate the growth of dendrites simply by applying and releasing pressure, causing the dendrites to zig and zag in perfect alignment with the direction of the force.
Applying mechanical stress to the solid electrolyte doesn’t eliminate the dendrite formation, but can control the direction of its growth. Instead of allowing them to cross from one electrode to the other, the MIT researchers proved they could be directed to remain parallel to the two electrodes. Thus, they remain harmless, allowing the solid-state cells to endure many charge-discharge cycles without shorting out.
Having demonstrated the theoretical principles of this process, the team will try to apply them to a functional battery prototype. Although a patent has been filed, the MIT researchers don’t intend to commercialize the system themselves. Instead, they think the companies working on the development of solid-state batteries should benefit from their findings. Hopefully, this will lead to putting solid-state batteries on the market as soon as possible.