CLEPA proposed this December 6 that renewable fuels can make combustion engines live longer and save jobs. Volkswagen thinks ethanol can deliver a carbon-neutral future in developing countries. These are hopes that do not stand a simple reality check like T&E (Transport & Environment) has performed with e-gasoline. Synthetic or not, renewable or not, the issue with burning stuff goes beyond carbon dioxide.
T&E asked IFPEN (IFP Energies Nouvelles) to perform the study. Interestingly, the testing company could not purchase the e-fuel: it had to produce it to make the tests. There’s no e-gas for sale nowadays. With the test results, we may never see them in fuel stations.
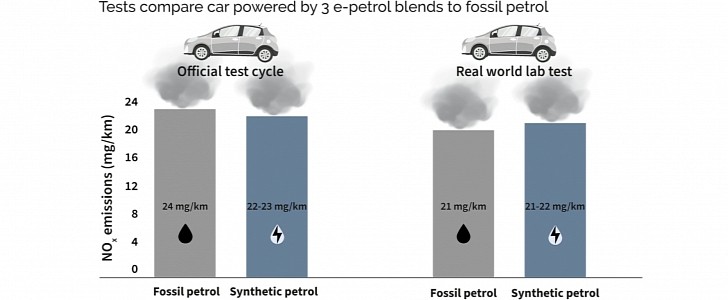
IFPEN created three sorts of e-fuel. Two different e-gasoline blends and one mix of e-petrol with 10% of second-generation ethanol (produced from biomass). This last e-gas would be comparable to the E10 used in Europe. A Mercedes-Benz A180 burned these fuels in WLTP and RDE testing cycles.
Although carbon emissions could be considered neutral thanks to their renewable origin (the carbon was extracted from the atmosphere), all other pollutants were still there. IFPEN discovered that e-gasoline emits as much nitrogen oxides as gasoline. The various types of NOx cause most breathing diseases in big cities and are highly poisonous. It was because of NOx that Dieselgate was such a scandal to the Volkswagen group. We would not doubt that engines burning pure hydrogen would also produce them.
The interesting bit of the research is that synthetic fuels generated three times more carbon monoxide than regular gasoline. Carbon monoxide is highly toxic: it combines with hemoglobin and prevents people from getting oxygen while breathing. In the WLTP cycle, it was three times higher. In the RDE cycle, it was “only” 1.2 to 1.5 times higher than with fossil fuel.
Things keep getting nasty for synthetic fuels. Ammonia levels are twice as much with two e-gasoline blends. Hydrocarbon emission is similar to that of gasoline in the RDE cycle and 23% to 40% lower, which may seem to be the only good news for these e-gasolines. However, it had one more advantage: a lower level of particulates more extensive than 10 nm. The really fine particulates below 10 nm were still there, in the same quantities as those produced by regular gasoline.
The T&E study shows that the problem with combustion engines is not restricted to the origin of the fuel they burn. Their very nature makes them a thing of the past. Apart from being extremely inefficient, they will also generate pollutants by burning fuel.
In that sense, avoiding increasing carbon levels in the atmosphere is just part of the solution and a very limited one. Big cities crowded with cars may still see high levels of ozone, ammonia, hydrocarbons, carbon monoxide, and a long etcetera of poisonous substances with e-fuels. Their origin is the combustion process, simply put.
If the idea is to use synthetic fuels, they must either help keep classic cars running or be used as a better way to store and carry hydrogen. Imagining they can help save the combustion engine for future generations is a big mistake that T&E helped eradicate with its study.
IFPEN created three sorts of e-fuel. Two different e-gasoline blends and one mix of e-petrol with 10% of second-generation ethanol (produced from biomass). This last e-gas would be comparable to the E10 used in Europe. A Mercedes-Benz A180 burned these fuels in WLTP and RDE testing cycles.
Although carbon emissions could be considered neutral thanks to their renewable origin (the carbon was extracted from the atmosphere), all other pollutants were still there. IFPEN discovered that e-gasoline emits as much nitrogen oxides as gasoline. The various types of NOx cause most breathing diseases in big cities and are highly poisonous. It was because of NOx that Dieselgate was such a scandal to the Volkswagen group. We would not doubt that engines burning pure hydrogen would also produce them.
The interesting bit of the research is that synthetic fuels generated three times more carbon monoxide than regular gasoline. Carbon monoxide is highly toxic: it combines with hemoglobin and prevents people from getting oxygen while breathing. In the WLTP cycle, it was three times higher. In the RDE cycle, it was “only” 1.2 to 1.5 times higher than with fossil fuel.
Things keep getting nasty for synthetic fuels. Ammonia levels are twice as much with two e-gasoline blends. Hydrocarbon emission is similar to that of gasoline in the RDE cycle and 23% to 40% lower, which may seem to be the only good news for these e-gasolines. However, it had one more advantage: a lower level of particulates more extensive than 10 nm. The really fine particulates below 10 nm were still there, in the same quantities as those produced by regular gasoline.
The T&E study shows that the problem with combustion engines is not restricted to the origin of the fuel they burn. Their very nature makes them a thing of the past. Apart from being extremely inefficient, they will also generate pollutants by burning fuel.
In that sense, avoiding increasing carbon levels in the atmosphere is just part of the solution and a very limited one. Big cities crowded with cars may still see high levels of ozone, ammonia, hydrocarbons, carbon monoxide, and a long etcetera of poisonous substances with e-fuels. Their origin is the combustion process, simply put.
If the idea is to use synthetic fuels, they must either help keep classic cars running or be used as a better way to store and carry hydrogen. Imagining they can help save the combustion engine for future generations is a big mistake that T&E helped eradicate with its study.