New promising battery chemistry called lithium-sulfur will replace the old Li-Ion batteries everyone is using today. Scientists at the University of Michigan have announced a breakthrough that allows Li-S batteries to be commercially viable.
Lithium-sulfur batteries are old-time friends to scientists but are yet to appear in commercial applications. Although they can hold much more energy than their Li-Ion counterparts, they have some problems that make them unstable and also unusable after only a handful of charging cycles. Now, scientists at the University of Michigan discovered a way to greatly extend Li-S batteries’ life while also making them safe.
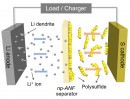
For that, they developed a biologically-inspired membrane made out of recycled kevlar that can isolate the lithium ions on the anode part from the polysulfides at the cathode. The same membrane can prevent the dendrites that form on the anode to puncture through to the cathode, a common cause of fires in traditional batteries.
Previously, the team behind the breakthrough used aramid infused with an electrolyte gel to stop the dendrites from reaching the cathode. But lithium-sulfur batteries have another problem - small molecules of lithium and sulfur flow to the lithium, attaching themselves and reducing the battery’s capacity. To solve this problem, they developed a membrane that simultaneously allows lithium ions to flow from the lithium to the sulfur and back—and to block the lithium and sulfur particles, known as lithium polysulfides.
“Inspired by biological ion channels, we engineered highways for lithium ions where lithium polysulfides cannot pass the tolls,” explains Ahmet Emre, a postdoctoral researcher in chemical engineering and co-first author of the paper in Nature Communications.
Thanks to this breakthrough, Li-S battery can get close to its full potential in terms of capacity and number of cycles. It’s also immune to extreme temperatures associated with automotive applications, such as those in cold winters and hot summers. The team at the University of Michigan believes the breakthrough will enable real-world batteries to withstand more than 1,000 cycles under fast charging.
According to a survey, American people expect a battery-electric vehicle to have at least 500 miles of range before considering buying one. Right now, only the Lucid Air fulfills this requirement, but soon this will be dwarfed by the new technologies in development. A battery having five times the capacity of a normal Li-Ion battery and able to withstand 1,000+ cycles would surely change the way electric vehicles are perceived.
Not long ago, a Tesla Model S with an experimental battery traveled 752 miles on a charge. While this is impressive, there’s little information about the battery’s life expectancy or the number of cycles it can endure before being unusable. Nevertheless, we see battery technologies pushing the boundaries of what’s acceptable for an electric car.
For that, they developed a biologically-inspired membrane made out of recycled kevlar that can isolate the lithium ions on the anode part from the polysulfides at the cathode. The same membrane can prevent the dendrites that form on the anode to puncture through to the cathode, a common cause of fires in traditional batteries.
Previously, the team behind the breakthrough used aramid infused with an electrolyte gel to stop the dendrites from reaching the cathode. But lithium-sulfur batteries have another problem - small molecules of lithium and sulfur flow to the lithium, attaching themselves and reducing the battery’s capacity. To solve this problem, they developed a membrane that simultaneously allows lithium ions to flow from the lithium to the sulfur and back—and to block the lithium and sulfur particles, known as lithium polysulfides.
“Inspired by biological ion channels, we engineered highways for lithium ions where lithium polysulfides cannot pass the tolls,” explains Ahmet Emre, a postdoctoral researcher in chemical engineering and co-first author of the paper in Nature Communications.
Thanks to this breakthrough, Li-S battery can get close to its full potential in terms of capacity and number of cycles. It’s also immune to extreme temperatures associated with automotive applications, such as those in cold winters and hot summers. The team at the University of Michigan believes the breakthrough will enable real-world batteries to withstand more than 1,000 cycles under fast charging.
According to a survey, American people expect a battery-electric vehicle to have at least 500 miles of range before considering buying one. Right now, only the Lucid Air fulfills this requirement, but soon this will be dwarfed by the new technologies in development. A battery having five times the capacity of a normal Li-Ion battery and able to withstand 1,000+ cycles would surely change the way electric vehicles are perceived.
Not long ago, a Tesla Model S with an experimental battery traveled 752 miles on a charge. While this is impressive, there’s little information about the battery’s life expectancy or the number of cycles it can endure before being unusable. Nevertheless, we see battery technologies pushing the boundaries of what’s acceptable for an electric car.