For what it’s worth, it seems humanity has overcome many of the hurdles standing in the way of mass adoption of electric cars. People trust them now, there are enough charging stations to go around, and range anxiety is all but gone, as most EVs now on the market come with more than decent capabilities. Yet, one major obstacle remains: charging times.
Despite the many fast charging options presently out there, how electric car batteries are made still makes it impossible even to come close to the time one needs to fill up the tank of an ICE car. So no matter what type of EV one drives, a significant amount of time spent allowing it to juice up must be taken into account ahead of any journey.
Strides are, of course, made to redesign batteries in such a way as to cut down those times, but the reality is we’ll probably not get anywhere in this respect for possibly decades to come.
Yet change has to start somewhere, and a possible starting point could be the solution a team of scientists from the Harvard John A. Paulson School of Engineering and Applied Science (SEAS) is working on.
Their solution is, of course, centered on solid-state batteries, the “holy grail for battery chemistry because of its high capacity and energy density,” as associate professor of materials science Xin Li said this week for The Harvard Gazette.
Li’s team claims to have created a stable lithium-metal battery that has the capability of being charged and discharged at least 10,000 times before becoming useless—for reference, a 100 kWh battery found in present-day EVs can provide at most 2,000 of these cycles.
Such a long-lasting battery would mean a car could be used for up to 15 years before in need of new hardware, but more importantly, thanks to the density, a full charge could be achieved in as little as 10 minutes.
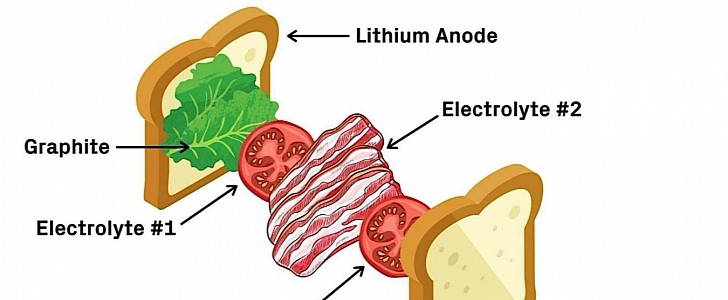
The secret behind the capabilities of the Harvard battery (we’re not told the hardware’s capacity) is its sandwich-like construction. Unlike current cell-based batteries, this one places the elements one on top of the other: the lithium metal anode, a coating of graphite, the first electrolyte, the second electrolyte, and finally, the cathode. Kind of like the bacon, lettuce, and tomato sandwich, Li adds.
For now, the researcher does not say how advanced this research is, and says the idea presented in Nature is for now just proof-of-concept.
Strides are, of course, made to redesign batteries in such a way as to cut down those times, but the reality is we’ll probably not get anywhere in this respect for possibly decades to come.
Yet change has to start somewhere, and a possible starting point could be the solution a team of scientists from the Harvard John A. Paulson School of Engineering and Applied Science (SEAS) is working on.
Their solution is, of course, centered on solid-state batteries, the “holy grail for battery chemistry because of its high capacity and energy density,” as associate professor of materials science Xin Li said this week for The Harvard Gazette.
Li’s team claims to have created a stable lithium-metal battery that has the capability of being charged and discharged at least 10,000 times before becoming useless—for reference, a 100 kWh battery found in present-day EVs can provide at most 2,000 of these cycles.
Such a long-lasting battery would mean a car could be used for up to 15 years before in need of new hardware, but more importantly, thanks to the density, a full charge could be achieved in as little as 10 minutes.
The secret behind the capabilities of the Harvard battery (we’re not told the hardware’s capacity) is its sandwich-like construction. Unlike current cell-based batteries, this one places the elements one on top of the other: the lithium metal anode, a coating of graphite, the first electrolyte, the second electrolyte, and finally, the cathode. Kind of like the bacon, lettuce, and tomato sandwich, Li adds.
For now, the researcher does not say how advanced this research is, and says the idea presented in Nature is for now just proof-of-concept.